Видео с ютуба European Pharmacopeia

European Pharmacopeia 11th edition effective January 2023

European and American Pharmacopoeia to Define Quality and Facts of NBCD’s

Presentation of the EDQM and its activities

Европейская фармакопея (EP/Ph.Eur.) пояснила

European pharmacopoeia

New Standards for UK Medical Cannabis from July 1st 2024(European Pharmacopoeia)

European Pharmacopoeia Ph. Eur.

Compliant with European Pharmacopoeia Chapter 2.1.7. on Balances

European Pharmacopeia - general

Biologicals in the European Pharmacopoeia – From vaccines to cutting-edge innovations

Validation of a European Pharmacopoeia method for quantification of a major grass pollen allergen

European Pharmacopoeia and complete TOC oxidation for PW and WFI

HPLC analysis of drugs according to pharmacopoeia

The Changing Face of Europe and the Implications of Brexit to the Pharma Industry

The Official Monograph for Cannabis Flower Joins the European Pharmacopeia - CannaReporter #Shorts
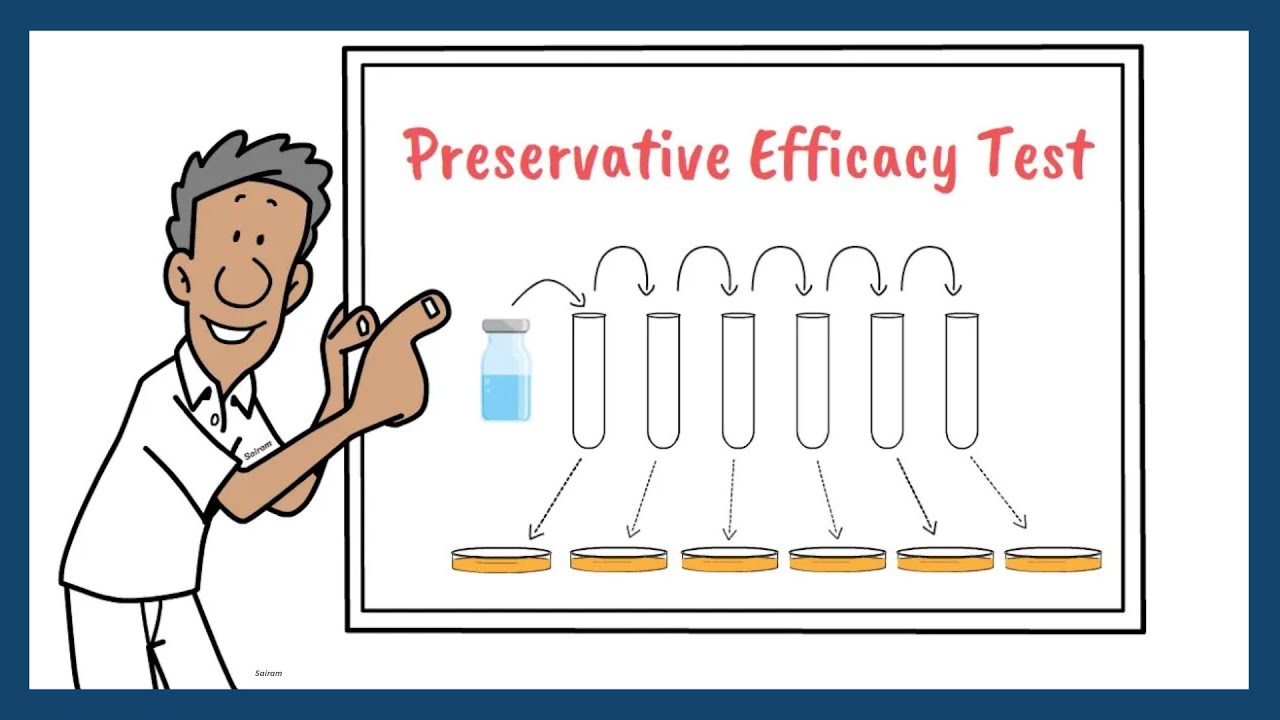
Preservative Efficacy Test | European Pharmacopoeia | Pharmaceutical Microbiology

Overcoming the challenges in new European Pharmacopoeia chapter for WFI production

EDQM, 50 years of leadership in the quality of medicines: paving the way for the future

Characterisation of non-compendial reference standards for impurities: How good is good enough?
The 9th Edition European Pharmacopoeia: Maintaining high quality standards in a dynamic environment